Chemical and Physical Changes and Properties
Physical changes are changes to a substance that does not change the type of matter.
Examples: cutting, grinding, polishing, boiling, melting, freezing
Chemical changes, on the other hand, are changes that change the type of matter.
These changes are usually observed during a chemical reaction, where one or more substances react to form one or more new substances (products). These changes are based on reactivity.
Examples: rusting, combusting, burning, cooking, decomposing, digesting
Examples: cutting, grinding, polishing, boiling, melting, freezing
Chemical changes, on the other hand, are changes that change the type of matter.
These changes are usually observed during a chemical reaction, where one or more substances react to form one or more new substances (products). These changes are based on reactivity.
Examples: rusting, combusting, burning, cooking, decomposing, digesting
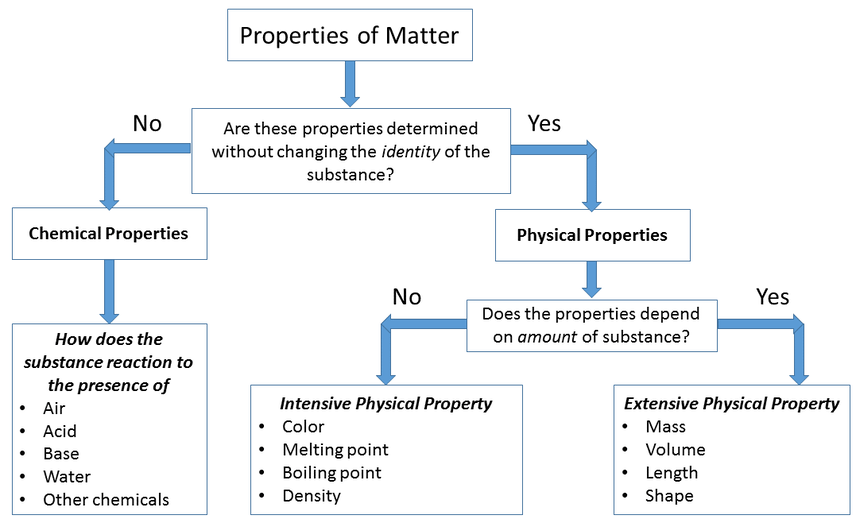
Image obtained from Chemistry LibreTexts: click on the button below to link to website.
